Cesium fluoride, therefore, is not Cs 2 +F 2− because the energy cost of forming the doubly charged ions would be greater than the additional lattice energy that would be gained. Not only is an electron being added to an already negatively charged ion, but because the F − ion has a filled 2 p subshell, the added electron would have to occupy an empty high-energy 3 s orbital. Na+(g) + Cl-(g) NaCl(s) Ho -787.3 kJ/mol Born Haber cycle for NaCl: Lattice energy cannot be measured empirically, but it can be calculated using electrostatics or estimated using the Born-Haber cycle. Furthermore, forming an F 2− ion is expected to be even more energetically unfavorable than forming an O 2− ion. Lattice energy is defined as the energy required to separate a mole of an ionic solid into gaseous ions. To form the Cs 2 + ion from Cs +, however, would require removing a 5 p electron from a filled inner shell, which calls for a great deal of energy: I 2 = 2234.4 kJ/mol for Cs. Q- If the formation of ionic lattices containing multiply charged ions is so energetically favorable, why does CsF contain Cs + and F − ions rather than Cs 2 + and F 2− ions?Ī- If we assume that U for a Cs 2 +F 2− salt would be approximately the same as U for BaO, the formation of a lattice containing Cs 2 + and F 2− ions would release 2291 kJ/mol (3048 kJ/mol − 756.9 kJ/mol) more energy than one containing Cs + and F − ions. Determine the lattice energy for NaCl by using the Born-Lande formula. Although the internuclear distances are not significantly different for BaO and CsF (275 and 300 pm, respectively), the larger ionic charges in BaO produce a much higher lattice energy. Find the Lattice Energy of NaCl (s) by the Born-Haber cycle method. Lattice energies are directly proportional to the product of the charges on the ions and inversely proportional to the internuclear distance. Calculate the enthalpy of the solution of NaCl(s). 
The relationship between the lattice energy. If the first four terms in the Born–Haber cycle are all substantially more positive for BaO than for CsF, why does BaO even form?Ī- The answer is the formation of the ionic solid from the gaseous ions. Click hereto get an answer to your question The lattice energy of NaCl(s) is - 790 kJ.mol-1 and enthalpy of hydration is - 785 kJ.mol-1. Following this convention, the lattice energy of NaCl would be +786 kJ/mol. Even though adding one electron to an oxygen atom is exothermic ( EA 1 = −141 kJ/mol), adding a second electron to an O −(g) ion is energetically unfavorable ( EA 2 = +744 kJ/mol)-so much so that the overall cost of forming O 2−(g) from O(g) is energetically prohibitive ( EA 1 + EA 2 = +603 kJ/mol). Q- Forming gaseous oxide (O 2−) ions is energetically unfavorable. The Born–Haber Cycle Illustrating the Enthalpy Changes Involved in the Formation of Solid Cesium Fluoride from Its Elements: Q- Arrange NaCl, MgS, AlN, and KBr in order of increasing lattice energy.Ī- The order of increasing lattice energy is KBr < NaCl < MgS < AlN. Q- Arrange GaP, BaS, CaO, and RbCl in order of increasing lattice energy.Ī- The order of increasing lattice energy is RbCl < BaS < CaO < GaP. It is derived by the charges on both ions upon the distance between the two charged atoms in the ionic compound. The NaF distance in NaF, which has the same structure as KF, is 231 pm. 
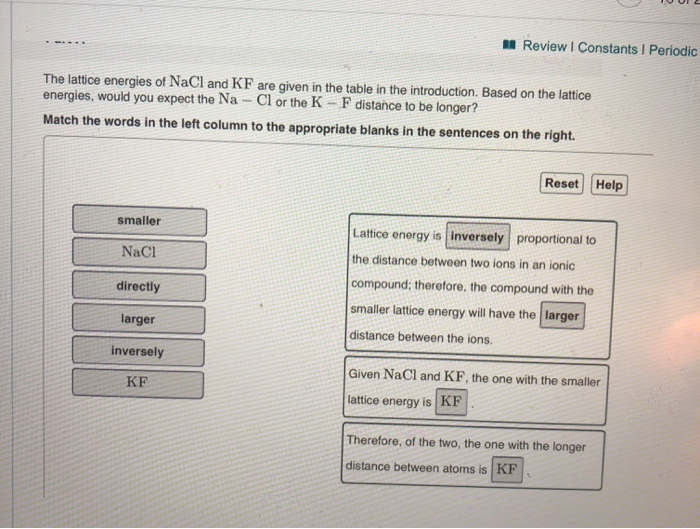
The four ionic compounds are N a C l, N a F, K B r, and K C l. The energies are found to be 9 2 2 k J / m o l, 7 6 9 k J / m o l, 7 1 8 k J / m o l and 6 8 8 k J / m o l respectively. That means that we will have to use theoretical values of their lattice enthalpies.Lattice energy is the most important factor in determining the stability of an ionic compound. Hint: lattice energy of any compound is the energy that is required to completely separate the respective ions in an ionic compound that form their respective gaseous states. The lattice energy of KF is 794 kJ/mol, and the interionic distance is 269 pm. The lattice energy of four ionic compounds W, X, Y and Z are measured. Let's look at this in terms of Born-Haber cycles of and contrast the enthalpy change of formation for the imaginary compounds MgCl and MgCl 3. It turns out that MgCl 2 is the formula of the compound which has the most negative enthalpy change of formation - in other words, it is the most stable one relative to the elements magnesium and chlorine. Using these values, UNaCl 867 kJ/mol, which is too negative (cf., 786.8 kJ/mol from Born. Lattice Energy (U) The amount of energy needed to fully separate one mole of an ionic solid into its gaseous ions is known as lattice energy (U). The question arises as to why, from an energetics point of view, magnesium chloride is MgCl 2 rather than MgCl or MgCl 3 (or any other formula you might like to choose). Calculating U for NaCl For a mole of ion pairs (N), using the Madelung constant (M), the expression for the potential energy of an NaCl-type lattice due to Coulombic interactions is L For the NaCl-type lattice M 1.74756, and for NaCl ro 280 pm. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
